This type of battery has been around for over a hundred years and is a well-proven technology. These batteries are characterized by their high power, i.e. they can deliver a high current in relation to their energy content.
Lead and sulfuric acid are harmful to humans and animals, so it is important to return used batteries for recycling.
There are several different types of batteries based on lead-acid chemistry.
All lead-acid batteries have
- A negative terminal which is made of spongy lead to get as large a surface as possible against the electrolyte.
- A positive terminal made of lead dioxide.
- An electrolyte consisting of liquid sulfuric acid mixed with water.
The image below shows the chemical processes during the charging and discharging of a lead-acid battery.

Charging of lead-acid batteries
When the battery is charged, water in the electrolyte is used to form sulfuric acid using sulfur sulfate in both the negative and positive terminals. A sub-step in this conversion creates pure hydrogen at the positive terminal.
When the charging process works perfectly, all the hydrogen is used to form sulfuric acid with the help of sulfur sulfate from both terminals. Unfortunately, the process is not always perfect and some hydrogen can be released and leave the battery, especially if the charging current is too high. It happens when the charging voltage gets too high.
This is why battery boxes for lead-acid batteries should always be closed with sufficient ventilation out into the open air.
Discharge of lead acid
When the battery is discharged, the sulfuric acid of the electrolyte is used to form lead sulfate in both the negative and positive terminals. When sulfuric acid is used, pure hydrogen and oxygen are formed at the positive terminal in one phase of the process. Later this hydrogen and oxygen create water that dilutes the electrolyte.
Unfortunately, the process is not always perfect and some hydrogen can be released and leave the battery, especially if the current is too high.
This is why battery boxes for lead-acid batteries should always be closed with sufficient ventilation out into the open air.
Battery construction lead-acid
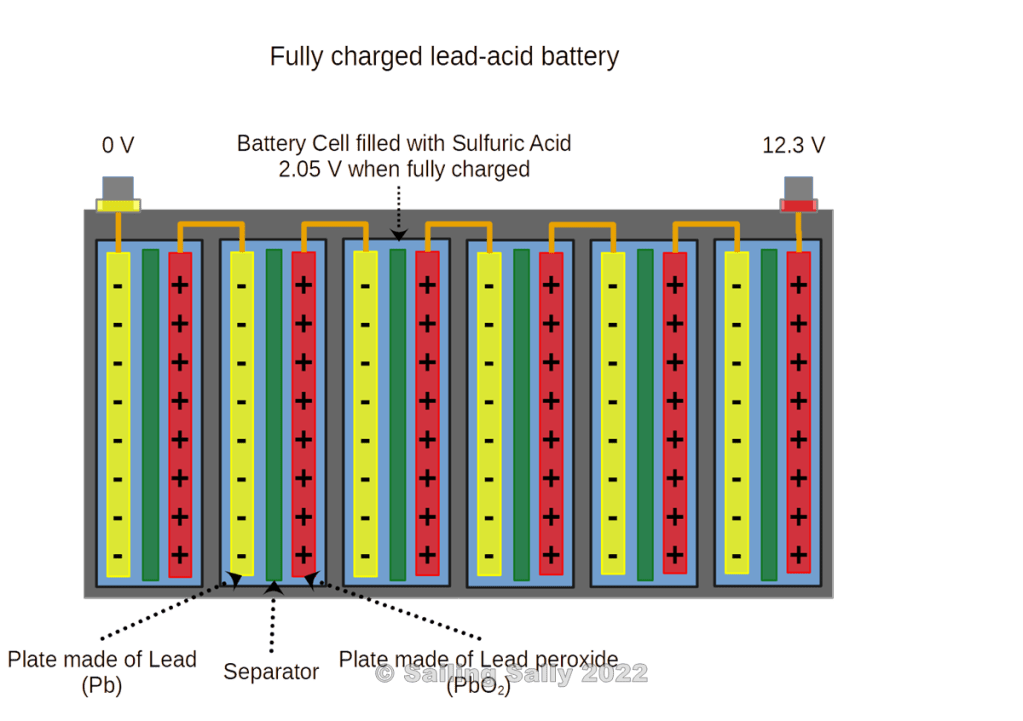
Lead-acid batteries are often built with a nominal voltage of 12V. Since a lead-acid battery cell gives 2.05V when it is fully charged, the battery contains 6 battery cells connected in series where each cell consists of its own container with anode and cathode immersed in sulfuric acid mixed with water. A fully charged lead-acid battery has a voltage of 12.3V. Immediately after charging, the voltage is higher, but after a few hours of rest, the voltage should be 12.3V.
Variations of the electrolyte in lead-acid
In principle, there are three different types of batteries like this and the difference is the composition of the electrolyte.
Flooded
The electrolyte is liquid. These batteries release hydrogen gas and also a little sulfuric acid which causes metal near the battery to corrode, e.g. connected cables. These batteries are not damaged if hydrogen gas is formed in them. Sealed batteries of this type are manufactured with a certain amount of extra electrolyte and this allows them to withstand some gassing before they become unusable. Open batteries can easily be filled with more water.
AGM
The electrolyte is bound in a fiberglass mat. These batteries are not damaged if hydrogen gas is formed in them, on the other hand, the water is used up and because they are prepared with a certain amount of extra water, they can withstand a certain amount of so-called gassing before they become unusable.
GEL
The electrolyte is in the form of a silicon gel. These batteries are sensitive to the formation of hydrogen gas which forms bubbles in the gel and causes permanent damage to the battery. This is why these batteries should not be used where large currents may be required, e.g. starting or bow thrusters.
Lead-acid battery container construction
The enclosure for the cells of lead-acid batteries can have different designs and basically, they differ in the following manner
Open or closed lead-acid battery cell
An open battery can be filled with additional electrolyte, actually distilled water because it is the water that disappears when the battery gasses during strong current when charging or discharging the battery.
It is not possible to add more electrolyte to a sealed battery. AGM and GEL batteries are always sealed.
Ventilated or unventilated
An unventilated battery has a free outlet for formed gases. Open cells are always unvented.
A vented (VRLA) battery has a safety valve that releases formed gas when the pressure becomes too high. AGM and GEL batteries are always ventilated and built so that as much of the hydrogen as possible is used (recombination), which reduces hydrogen emissions by up to 99% compared to flooded batteries.
Design of battery terminals
By varying the design of the anode and cathode, there are batteries with slightly different properties which means that they can, for example, deliver greater current, have more charge cycles, or withstand deep discharge better.
Form of battery cells
There are square (prismatic) or round (circular) battery cells.
With square cells, the energy density can be increased when the outer battery box is square. Round battery cells are often easier to manufacture because long battery terminals are rolled together instead of connecting several in parallel as in prismatic cells.
Most 12V lead-acid batteries have prismatic battery cells.
The thickness of lead plates
More and thinner plates in the battery terminals mean that the battery gets more power in relation to its capacity. The disadvantage is that the battery becomes more sensitive to deep discharge and can receive fewer charge cycles.
Fewer and thicker plates mean that the battery has lower power in relation to its capacity. The advantage is that it withstands deep discharge better and gets more charge cycles.
Chemical Variations
By varying the chemistry in the battery poles, you can create lead-acid batteries with better properties.
Lead-Calcium batteries
In order to keep the lead in place in the plates of the battery terminals, a net made of an alloy containing antimony is usually used. When you replace the antimony alloy with a calcium alloy, you get batteries that form less gas and thus consume less electrolyte. They also have less self-discharge.
There are variations on this construction where silver has also been mixed into the calcium alloy, resulting in an even lower degree of self-discharge and greater resistance to sulphation.
These batteries are still most suitable for use as starter batteries as they still have relatively poor resistance to deeper discharge and relatively few charge cycles.
Lead-Carbon batteries
This is a fairly new type of lead-acid battery where a carbon-based structure has been added to the battery’s negative pole (anode). This means that the current is distributed more evenly in the battery pole, resulting in batteries that
- has better tolerance to partial discharge and partial charge resulting in more charge cycles
- can be discharged more
- can be charged faster
- gets less sulfation on the negative pole
These batteries work well as service batteries as they can withstand relatively deep discharges and have many charge cycles even from slightly deeper discharges.
Problem
Stratification
This does not apply to GEL or AGM batteries.
When a battery is charged, sulfuric acid is formed which is heavier than the electrolyte itself, which is mixed with water. Some of the sulfuric acid does not mix with the rest of the electrolyte and will collect at the bottom of the battery. This reduces the capacity of the battery. You can make the sulfuric acid mix up by stirring the electrolyte in some way, e.g. shaking the battery or overcharging it so that gas bubbles are formed and stir the electrolyte.
Shedding
During discharge, the material in the positive battery pole is converted into a material with a slightly larger volume, which causes the battery pole to loosen up a little with each charge cycle. Over time, this causes the battery terminal to break.
Corrosion of battery terminals
The supporting grid in the positive battery terminal corrodes at the end of the charging period because the sulfuric acid content is highest then. Over time, this causes the battery terminal to break.
Sulphation
When the battery is discharged, lead sulfate forms on the negative battery terminal and if the battery remains discharged for a long time, the substance forms crystals that prevent charging. This reduces the battery’s ability to store electrical energy.
Care of lead-acid batteries
- Charge the batteries with a 3-step charger and make sure to keep them charged when you leave the boat. If the battery is not fully charged, it sulfates and loses capacity.
- Keep the battery clean and remove oxide from the battery terminals.
- If you are going to store a lead-acid battery, make sure it is fully charged and recharge it periodically, otherwise, it will sulfate.
Connection of lead batteries
Since a manageable lead-acid battery (60 kg) can contain approximately 1200 Wh of available energy, it may be necessary to connect several batteries in a battery bank to get enough energy.
This amount is not enough for the consumption of the fictitious sailboat in these articles, see Electrical system, Part 3 – Battery selection in the subsystems where there is a calculation of the energy requirement on board.
If the boat needs to store more energy, just start connecting batteries in parallel or in series, see article Electrical system, Part 3 – Connect the battery bank.
Keep in mind that
- Only connect batteries of the same capacity and age from the same supplier.
- Do not connect more batteries in parallel than the manufacturer prescribes.
- Do not connect more batteries in series than the manufacturer prescribes.
- Before connecting the batteries, make sure the batteries are charged and have the same resting voltage.
Which lead-acid batteries should you choose?
Only use sealed batteries, this means that sulfuric acid cannot leak into the bilge. Sulfuric acid is corrosive and destroys metal it comes into contact with, e.g. grommets and keel bolts. It is also dangerous for humans.
In Electrical system, Part 3 – Battery selection in the subsystems you can read more about the reasons for the battery selection in the boat used as a reference in the articles.
Protective functions
Remember to see the batteries as part of a larger system that includes both chargers and consumers. To protect the batteries and the boat, you must ensure that the battery solution is monitored, i.e. that it turns off charging when the batteries are fully charged and that they are disconnected from consumers when they are about to be discharged. Chargers must know the battery temperature and actual charging voltage.
There must also be a protection function that disconnects the batteries when the current to or from them becomes too big. This is to prevent batteries or cabling from overheating.
If you choose to build yourself, I recommend that you
- Use automatic disconnection of the charger when the battery is fully charged.
- Use automatic disconnection/shutdown from the consumers when the voltage gets too low.
- Use automatic disconnection of the battery when the current to/from the battery becomes too great.

Du måste vara inloggad för att kunna skicka en kommentar.