This type of rechargeable battery has existed since the end of the 70s and is based on lithium ions that are stored in molecular layers in the positive and negative terminals of the battery. Hence the name lithium-ion batteries. Compared to lead batteries, they are lighter, charge faster, and have more charge cycles.
Lithium is harmful to humans and animals, so it is important to hand in used batteries for recycling.
There are several different types of lithium-ion batteries and they are named after the materials that are used in the positive terminal of the battery.
- The negative terminal usually consists of a graphite mix with a little silicon.
- The positive terminal consists of a layered metal oxide that can store lithium ions, e.g. iron phosphate as in LiFePO4 batteries.
- The electrolyte is liquid and contains lithium salt dissolved in an organic liquid and bound in paper. An organic liquid can start to burn if the temperature gets high enough. Research is therefore underway to develop a solid electrolyte without flammable substances. Such a battery is called a ”solid-state” battery.
Do you want even more facts about lithium-ion batteries? Then you can start with Wikipedia.
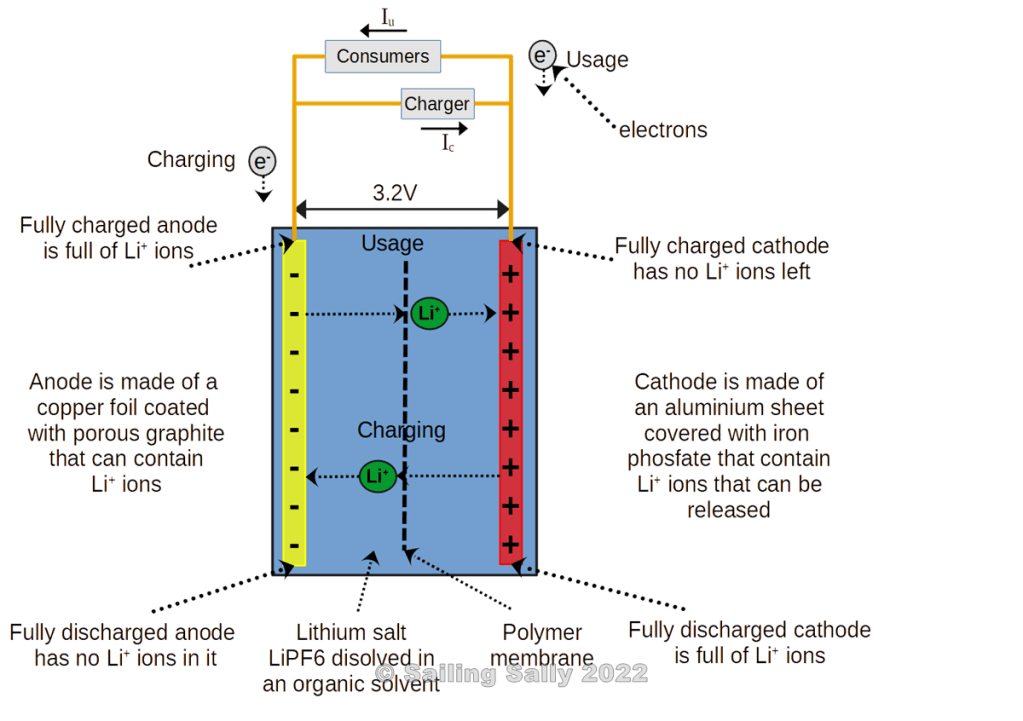
Lithium ion charging
When the battery is charged, lithium ions move from the positive terminal to the electrolyte and on to the negative terminal. The lithium ions are found in a layered structure in the positive terminal and are stored in the layered carbon structure of the negative terminal.
When the battery is completely discharged, there are no lithium ions left in the positive terminal.
The battery monitoring device must be able to equalize the voltage across all cells of the battery so that the voltage across each battery cell is the same. Ideally, there should only be a few thousandths of a volt difference between the cells. An ordinary voltmeter can possibly measure hundredths of a volt and is not enough to measure the cell voltage.
Lithium-ion discharge
When the battery discharges, lithium ions are released from the layers in the negative terminal into the electrolyte and are carried to the positive terminal where they are stored in the layered lithium structure of the positive terminal.
In a fully charged battery, all the lithium-ion slots on the positive terminal are occupied, while all the slots on the negative terminal are free.
This reversible process can be repeated without destroying the layered structures. In chemistry, the process is called intercalation and forms the basis of lithium-ion batteries.
Compared to lead-acid batteries, lithium batteries have several advantages
- They can store more energy per kg battery
- They receive a constant charging current
- They can be discharged to a greater degree
- They withstand many more charge cycles and still have about 80% of the capacity left after going through all the charge cycles.
Because they have different charging characteristics (different voltage levels), you cannot simply replace lead-acid batteries with Lithium-ion. Since the charging voltage is basically constant throughout the charging process, a shunt is also needed to measure the battery’s charge level.
Variations lithium-ion cells
Design of battery terminals
Form of battery cells
Just as with lead-acid batteries, there are square (prismatic) or round (circular) battery cells.
With square cells, the energy density can be increased when the outer battery box is square. Round battery cells are often easier to manufacture because long battery terminals are rolled together instead of connecting several in parallel as in prismatic cells.
Chemical Variations
By varying the chemistry in the battery terminals, you can create lithium-ion batteries with different properties.
The chemistry of the positive terminal is used to indicate the type of lithium-ion battery it is.
The negative terminal can also be made from different substances. So far, it is most common to use carbon in the negative terminal, and this limits the energy density to approx. 370 Wh/kg in the battery, regardless of the design of the positive terminal.
The table below shows some of the most common materials today and development is still ongoing.
| Abbreviation | Type of chemistry positive terminal | Characteristics | Usage |
| NCM | Lithium cobalt magnesium oxide | High energy density | cars |
| NCA | Lithium nickel cobalt aluminium oxide | High energy density, long lifetime | cars |
| LMO | Lithium magnesium oxide | High energy density | phones, cars |
| LFP | Lithium iron phosphate | Safe, no exotic metals | cars, boat |
| LCO | Lithium Cobalt oxide | High energy density | laptops |
Lithium-ion battery in boats
For use in boats, LFPs are recommended because they have a good lifespan and are the safest to use compared to the other types of lithium-ion batteries.
Battery build-up LFP
LFP batteries are often built with a nominal voltage of 12V or 24V. Since an LFP battery cell gives 3.2V when it is fully charged, a 12V battery contains 4 battery cells connected in series where each cell consists of its own container with electrolyte, anode, and cathode that are separated by a membrane that can let lithium ions through.

Problem
Unbalanced cell voltage lithium-ion batteries
Because lithium-ion batteries have an almost constant voltage throughout the charging and discharging process compared to a lead-acid battery, these batteries are much more sensitive to whether the cells in the battery have different voltages. The voltage difference between a 20% charged and a 95% charged battery is very small, about 0.1V, and this means that for every 10% charge the voltage changes by approximately 0.01V.
- When one of the cells has reached maximum voltage, the charging is stopped and if the cells are out of balance, the other cells will not be fully charged.
- When the battery discharges and a cell reaches the lowest allowable voltage, the entire battery is disconnected and the energy in the remaining cells cannot be used.
Lithium-ion battery care
There are some rules that you must always follow
- Do not discharge the battery too quickly
- Do not discharge the battery to much
- Do not charge when it is too cold or too hot
- Leave the battery charged to 70-80% when disconnected from the charger
If you want to increase the life of the battery further, you must make sure that
- Keep the battery cool, 15-20 degrees Celcius.
- Do not charge more than 95% SoC
- Do not discharge more than 25% SoC
Interconnection of lithium-ion batteries
Despite the fact that lithium-ion batteries have a higher energy density than lead-acid batteries, a 12V battery with a capacity of 400Ah weighs about 50 kg. It provides just over 300Ah (3600 Wh) of available energy and is not quite enough for the consumption of our fictitious sailboat, see Electrical system, Part 3 – Connect the battery bank where there is a calculation of the energy requirement on board.
If the boat needs to store more energy, you need to connect batteries in parallel. Make sure there is a BMS that can balance the batteries.
Which lithium batteries should you choose?
Only choose LFP batteries for the boat, they are the only lithium-ion batteries that are really safe today and I don’t know of any case where they have started a fire in a boat.
In Electrical system, Part 3 – Battery selection in the subsystems you can read more about the justification of the battery selection for the fictitious boat used as a reference in the articles.
Protective functions
Remember to see the batteries as part of a larger system that includes both chargers and consumers. In order to protect batteries and boats, you must ensure that the battery solution is monitored, i.e. that it stops charging when the batteries are fully charged and that they turn off the power outlet when they are about to reach the lowest charged level. Chargers must know battery temperature and actual charging voltage.
There must also be a function that disconnects the batteries when the current to or from them becomes too great. This is to prevent the batteries and electrical cables from overheating.
The battery monitoring must be able to equalize the voltage across all cells of the battery so that the voltage across each battery cell is the same. There should only be a few thousandths of a volt (0.003V) difference between all the cells in the battery. Between the batteries, in a battery bank, there should be a difference of a maximum of a few hundredths of a volt (0.03V)
Make sure that chargers and batteries can work together, e.g. if you have chargers from Mastervolt or Victron, it makes it easier to also buy batteries from the same manufacturer.
If you choose to build yourself, I recommend that you
- Uses 12V/24V batteries with built-in cell balancing.
- Use BMS for automatic balancing of parallel connected batteries.
- Build in automatic disconnection of the charger when the battery is fully charged.
- Build in automatic disconnection/shutdown from the consumers when the voltage gets too low.
- Build in automatic disconnection of the battery when the current to/from the battery becomes too great.
- Build in automatic switch-off when the temperature is too low.
- Build in automatic disconnection at too high a temperature.

Du måste vara inloggad för att kunna skicka en kommentar.