There are many different types of rechargeable batteries that can be used in boats. In this article, I go through how the most common types of batteries work, how they can be connected together and how they are charged for the best lifespan.
This subject area is comprehensive and I have split the article into several parts. This is the main article that ties it all together. There are seven more articles that describe different sub-areas and that’s it
- There are a lot of concepts that have to do with batteries and charging and in the article Electrical system, Part 3 – Battery terms I have described the ones used in these articles.
- You find more details about lead batteries in Electrical system, Part 3 – Facts about lead batteries.
- You find more information about lithium-ion batteries in Electrical system, Part 3 – Facts about lithium-ion batteries.
- A comparison of characteristics and costs of the battery types in Electrical system, Part 3 – Battery comparison.
- The reasons for the battery selection in the various subsystems in the fictitious sailboat, Electrical system, Part 3 – Battery selection in the subsystems
- For how to connect a battery bank with several batteries, see Electrical system, Part 3 – Connect the battery bank.
- More information on how battery chargers work and how they are connected can be found in the article Electrical system, Part 3 – Battery chargers.
The principle of rechargeable batteries
Rechargeable batteries in a sailboat are based on electrical energy being stored as chemical energy in the battery when it is being charged. When using the battery, the stored energy is converted back into electrical energy.
This sketch shows the principle of such a rechargeable battery. The battery consists of a negative terminal (anode) and a positive terminal (cathode). They are in the same electrically conductive substance (electrolyte).
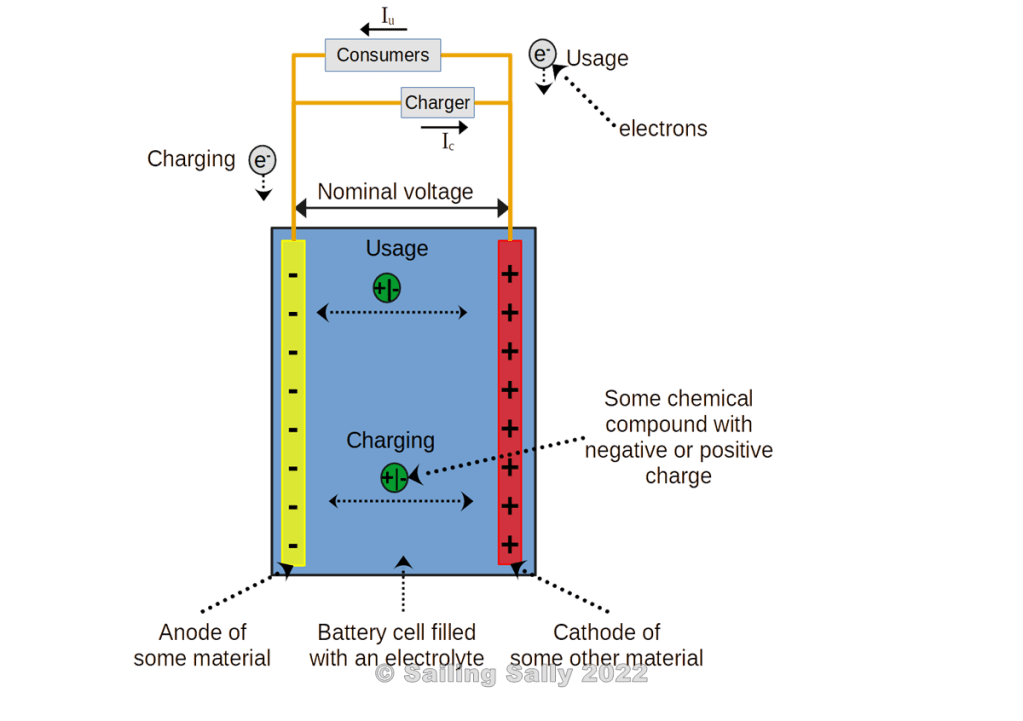
Charging
When a battery is charged, the voltage on the battery’s positive terminal is increased so that it becomes slightly higher than the battery’s resting voltage. Then a current begins to flow to the positive terminal of the battery. It starts a chemical reaction that uses the supplied electricity to convert the materials in the battery’s terminals into other substances, thus storing the supplied energy. When the charging voltage reaches a certain value, all the material is considered to be converted and the battery is considered fully charged. If the charging voltage is raised further, other chemical reactions can start. In most cases, the battery is damaged by excessive voltage.
Discharge
When the battery is used, the voltage on the positive terminal of the battery is lowered to below the resting voltage. Then a reverse chemical reaction starts which releases the stored energy in the form of an electric current from the positive terminal of the battery. In this chemical reaction, the battery terminals are converted back to the same material as before. When all the material is converted, the battery is completely discharged. If the voltage is lowered further, other chemical reactions can start. In most cases, the battery is damaged by low voltage.
It is these two processes that make rechargeable batteries possible.
Charging problems
Unfortunately, the processes are not always perfect and this means that the battery wears out more or less each time it is charged and discharged. There are certain things that cause a battery to wear out more and get a shorter lifespan. In principle, all rechargeable batteries are more or less sensitive to these things
- Incorrect charging voltage
- Incorrect charging process
- Charging current too large
- Draw too large current
- Charging voltage too high
- Charging voltage too low
- Discharge the battery too much
- High ambient temperature
- Low ambient temperature
Battery chemistry
Today mainly two different types of chemical processes are used in rechargeable batteries on boats, these are
- A chemical reaction between lead and sulfuric acid diluted with water, so-called lead-acid batteries. You can read more in Electrical systems, Part 3 – Facts about lead batteries.
- A chemical process called intercalation where lithium ions are stored in the battery terminals’ layered structures of lithium and graphite respectively, so-called lithium-ion batteries. You can read more in Electrical system, Part 3 – Facts about lithium-ion batteries.
Which batteries to choose?
How to know what type of battery to use and how much battery capacity will be needed?
The article Electrical system, Part 3 – Battery comparison contains a compilation of the characteristics of the different battery types and lifetime cost for energy storage.
In the article, Electrical system, Part 3 – Battery selection in the subsystems I write about the various factors that underlie the battery selection in the various electrical subsystems on board.
Connection of batteries
Often, more capacity is needed than is available in one battery, and this is solved by connecting several batteries in parallel. If the subsystem needs a higher voltage than what is in a battery, you solve it by connecting batteries in series.
In the article Electrical system, Part 3 – Connect the battery bank there is more to read about this.
Battery charger
Because the batteries we use in electrical systems on board are expensive, only use quality multi-stage automatic chargers designed for the type of batteries the boat has in its battery banks and ensure the chargers are properly installed and configured.
In the article Electrical system, Part 3 – Battery chargers you can read more about battery chargers and how they are connected.
In conclusion
There are several parameters that govern how to design the storage of electrical energy on a sailboat. The most important ones are
- Which electrical consumers are there?
- Where are the large consumers mounted on the boat?
- How much energy needs to be able to be stored?
- How much space is there for batteries and charging equipment?
- Are redundancy and backup needed when something breaks?
- How do different regulations for the installation of batteries and charging equipment affect the design?
- How much can it cost?
The answers to these questions will guide the design and produce different results depending on what the answers look like. This means that the overall construction can differ quite a lot from boat to boat in terms of the number and size of battery banks.
When it comes to the execution of the electrical installation, it must always follow the existing installation regulation and be done in the same way in all installations. The regulation exists to eliminate the risk of personal injury and fire due to faults in the electrical systems.


Du måste vara inloggad för att kunna skicka en kommentar.